
However, some proteins are highly elongated, either as a string of small globular domains or stabilized by specialized structures such as coiled coils or the collagen triple helix. Peptides larger than 50 kDa typically form two or more domains that are independently folded.
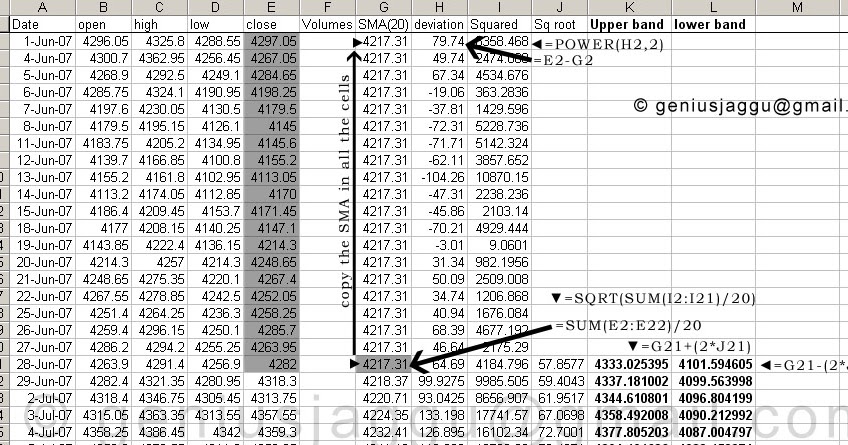
Peptides from 10 to 30 kDa typically fold into a single domain. Most proteins fold into globular domains, which have a minimal surface area.
Protein folding is driven largely by the hydrophobic effect, which seeks to minimize contact of the polypeptide with solvent. Most proteins fold into globular domains. A combination of hydrodynamics and electron microscopy is especially powerful. Finally, rotary shadowing and negative stain electron microscopy are powerful techniques for resolving the size and shape of single protein molecules and complexes at the nanometer level. The molecular weight can be determined by combining gradient sedimentation and gel filtration, techniques available in most biochemistry laboratories, as originally proposed by Siegel and Monte. It is recalled that a gel filtration column fractionates proteins on the basis of their Stokes radius, not molecular weight. Instead, a simple guideline is presented, based on the measured sedimentation coefficient and a calculated maximum S, to estimate if a protein is globular or elongated. Readers are reminded that the Perrin equation is generally not a valid approach to determine the shape of proteins. This review collects a number of simple calculations that are useful for thinking about protein structure at the nanometer level. Sedimentation and gel filtration are hydrodynamic techniques that can be used for this medium resolution structural analysis. An important part of characterizing any protein molecule is to determine its size and shape.
